By Catie Summers, V Form
How Do Candles Burn? A Chemistry Annotated Diagram
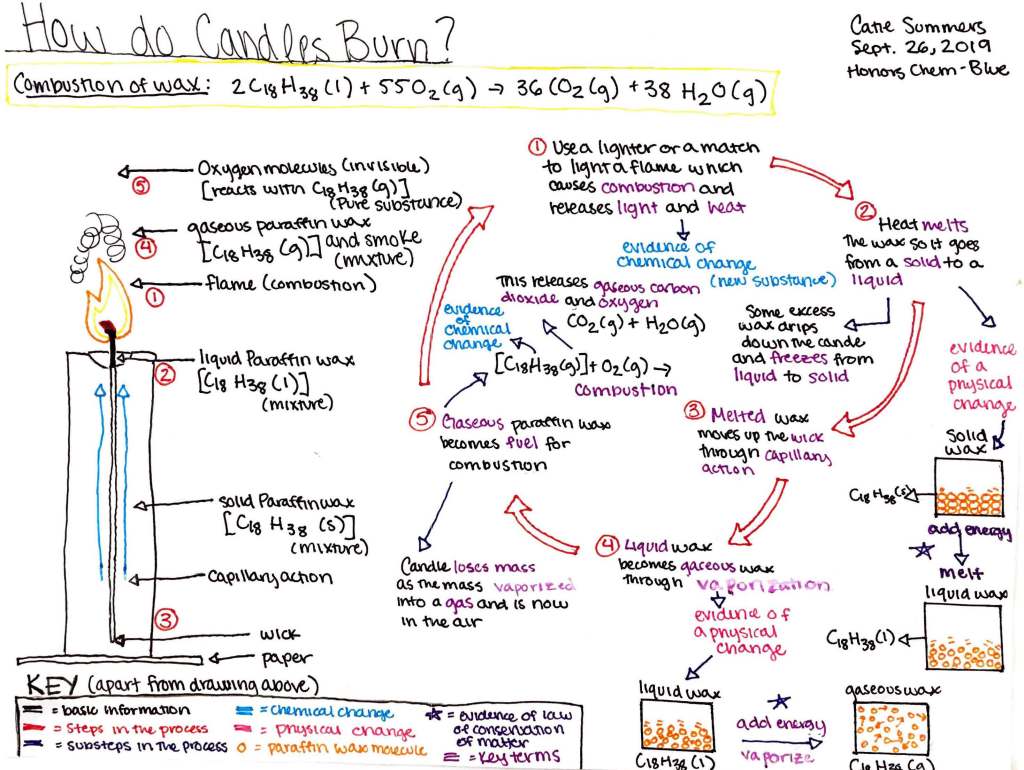
Teacher’s Note: In 1848, physicist Michael Faraday delivered a series of holiday lectures at the Royal Institution in London on the topic of “The Chemical History of a Candle.” During the lectures, Faraday used his observations of a burning candle as inspiration for relating, to a lay audience, a veritable encyclopedia of fundamental principles of physics and chemistry. In Honors Chemistry at St. Mark’s, we strive to recreate Faraday’s sense of wonder by performing simple experiments on a candle and interpreting the results in light, based on what we have learned about atoms, molecules, and chemical reactions. Students are challenged to determine the nature of a candle’s fuel and describe the process by which a candle’s flame perpetuates itself. In particular, Catie Summers’s eye-catching visual summary of this process reflects her efforts to link macro-scale observations with molecule-level interpretations.
Catie Summers is a V form boarding student from Amherst, New Hampshire. Her favorite classes are English and studio art.


