By Holden LeBlanc, Class of 2021, Burnett Prize Winner (20-21 School Year)
The George Hall Burnett in History is awarded on the basis of a special essay in American history.
Carolyn Markland, a grandmother from Jacksonville, Florida, was a lover of animals and spent years fostering rescue pets after retiring as an environmental engineer. Markland, however, struggled with back pain due to a degenerative disc disease for years. After trying different medications with little relief, a doctor prescribed Markland the fentanyl-based drug Subsys to subdue her pain. Markland took a dose of Subsys before going to sleep on July 2, 2014. When Markland’s daughter went to check on her mother the next day, she discovered her dead in her bed with a Subsys canister lying at her side. Although Markland’s overdose was the first death connected to Subsys, many more were looming. Along with thousands of others, Markland died from overdosing on prescription opioids, but to understand how this happened, it is essential to recognize the changes in the United States’ policy towards opioids.
Opium, the active ingredient in opioids, is a substance that blocks pain by stimulating the release of the chemical dopamine in the body. Opium is a naturally occurring substance found in poppy plants, which humans have cultivated for centuries seeking their medicinal effect. For most of America’s history, up until the mid-twentieth century, doctors utilized opioids like morphine and later heroin as a crude form of anesthesia for surgery and for managing debilitating pain. After soldiers who received these powerful opioids became addicted in the early twentieth century, however, the United States government banned heroin and severely limited morphine use in 1924. Throughout the first half of the twentieth century, opioid use in the U.S. remained relatively low, as much of the population viewed partaking in drug culture as morally wrong and deviant.
Attitudes began to change with the counterculture movement of the 1960s which embraced drugs such as marijuana and LSD. These drugs, however, were expensive, and after a large segment of the youth became drug dependent, many turned to cheaper and more potent alternatives, including heroin. The heroin epidemic was even worse among Vietnam Veterans due to the large supply of the drug in Southeast Asia, which grew vast amounts of the poppies used to make heroin.2 In response to the increase in drug use within the civilian and military population, President Richard Nixon declared a war on drugs in 1971. Initial efforts to stop the flow of drugs into the United States, however, provided little help. Between the years 1980 and 1988 alone, the number of heroin users increased by 750%.3 Because of the widespread abuse of opioids like heroin throughout the 1970s and 1980s, doctors seldom prescribed legal opioids such as Vicodin out of fear of addiction.
Public perception of opioids, however, shifted again during the 1990s in response to reports that cancer patients were denied opioids that would eliminate unnecessary pain from their treatments. Drug lobbyists used this surge of new public support to petition Congress to deregulate the opioid market.4 Drug companies vowed to create new, less addictive, but equally effective opioids to combat chronic pain. Leading this movement was Purdue Pharma, which developed the oxycodone pill named Oxycontin in 1996.
Purdue developed a timed-release mechanism for Oxycontin, as opposed to an instant delivery of opioids in the body. For this reason, Purdue marketed the drug as a safer alternative to traditional opioids. At first, doctors mainly prescribed the drug for patients suffering from terminal illnesses, but Purdue wanted Oxycontin more widely prescribed for managing all types of pain. This led Purdue to embark on a $208 million marketing campaign to convince doctors that Oxycontin was a safe and effective tool for managing pain. Purdue focused their marketing efforts on coal mine and steel manufacturing communities with large numbers of blue-collar workers suffering from chronic pain.5 Purdue’s marketing strategy proved remarkably successful, as doctors began prescribing Oxycontin for millions of users with varying levels of chronic pain. Despite these claims of low addiction rates, as prescription rates increased, opioid addictions followed. In response to rising concern over the addictive nature of Oxycontin, Purdue doubled down on its product. Purdue claimed that Oxycontin users who showed signs of addiction were not actually addicted but suffered from ‘pseudo-addiction,’ which meant that the medication was not properly treating their pain. Purdue advised doctors to increase the dosage of patients with these cases of ‘pseudo-addiction’ to fully alleviate the patient’s pain, maintaining that Oxycontin was safe with a low chance for ‘real’ addiction.6 In recent years, however, scientists have almost unanimously stated there is no proof behind the claims of pseudo-addiction by Purdue and that increasing the dosage of opioids like Oxycontin increases the risk for addiction.
Although scientists disproved Purdue’s claims of low addiction rates for Oxycontin and politicians categorized their marketing tactics as predatory, the damage that the drug caused remains. Between 1998, approximately when Purdue began its mass marketing campaign, and 2008, reported abuse of prescription opioids more than doubled. This large increase is extremely concerning considering the American Society of Addiction Medicine estimates that 23% of those who abuse prescription opioids become users of heroin.7 The CDC describes this movement from legally prescribed opioids to a surge of heroin use as the second wave of the opioid epidemic, beginning in 2010.8 This graph, however, displays an even more concerning trend that has quickly accelerated the severity of the opioid epidemic, which is the rise of deaths from chemically-altered synthetic opioids. In 2018 alone, synthetic opioid overdoses accounted for 31,000 deaths, contributing to 67% of all opioid-related fatalities that year.9 The majority of synthetic opioid deaths involved fentanyl, a highly addictive and dangerous synthetic opioid one hundred times more potent than morphine.10 It is a schedule II drug, the highest classification for legally prescribable drugs due to their potential for harming the patient and causing addiction.11 The drug has become a popular replacement in the illicit drug trade for heroin due to its lower price and immense potency.
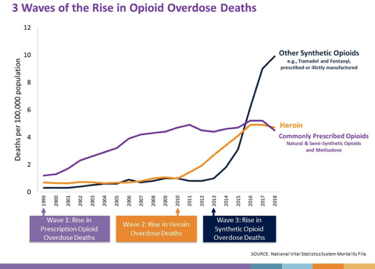
Since the increase in prescription opioid use in 1996, over 400,000 Americans have died from opioid overdoses. The CDC estimates that opioid misuse costs U.S. taxpayers $24 billion in medication costs and a further $75 billion in healthcare costs on a yearly basis.13 Although the American people have suffered greatly with the loss of hundreds of thousands of their loved ones, the companies that began and fueled the opioid epidemic have not received punishment or provided much compensation for the damage they caused. Federal and state prosecutors have opened thousands of cases against opioid manufacturers and distributors, only to settle lawsuits for fines making up a small portion of these company’s yearly revenues. Before 2020, no pharmaceutical CEO was successfully criminally charged for their part in the opioid epidemic.
It was not until one opioid manufacturer, Insys, took Purdue’s blitzkrieg marketing strategy to a new extreme that criminal prosecution succeeded, using a law originally meant to prosecute the mafia. The Racketeer Influenced and Corrupt Organizations Act has proven itself as an effective tool for criminally charging pharmaceutical executives who contributed to the Opioid Epidemic. By convicting CEOs and other executives and sending them to jail for engaging in a criminal conspiracy to push dangerous opioids that contributed to the deaths of thousands of Americans, the prosecution of Insys sends a strict message that the U.S. justice system will severely punish criminal behavior in pursuit of profits.
To begin the paper is a brief history of opioid manufacturer Insys Therapeutics and its founder John Kapoor. Additionally, this section details the illegal actions which Insys committed that perpetuated the opioid epidemic. Next is a description of the Racketeer Influenced and Corrupt Organizations Act, including its history and use in the corporate and pharmaceutical sectors. The paper’s final section describes how the Department of Justice successfully charged Insys executives with RICO violations for their illegal actions, and ultimately what these convictions mean for the entire legal opioid industry.
Selling Fentanyl at All Costs
Beginning in 2012, Insys Therapeutics displayed a blatant disregard for patients’ health in its reckless pursuit of profits. Founder of Insys, John Kapoor, became a billionaire through investing in the pharmaceutical industry. Kapoor was born in India to a working-class family.
After studying medicine in Mumbai, India, he received financial aid to move to the United States and attend the State University of New York at Buffalo, graduating in 1972.14 Kapoor started in the pharma industry as a plant manager at Invenex Laboratories in Grand Island, New York, where he met his future wife Editha.15 In 1978, Kapoor joined the sales team at generic drugmaker LyphoMed, where he began to show his incredible marketing prowess. But after almost a decade in the industry, Kapoor desired to run a pharma company of his own. Kapoor had negotiated a stipulation in his job contract that acknowledged he would be the first in line to buy LyphoMed should the owners want to sell the company. Kapoor got the opportunity to buy his former employer in 1981 for $2.7 million by taking several “loans from investors and a $24,000 second mortgage on his Evanston home.”16 Kapoor’s gamble paid off as Congress pushed forward legislation favoring generic medicine in the 1980s. This in turn increased sales from $19 million in 1983 to $159 million in 1989. LyphoMed, however, faced scrutiny from a Food and Drug Administration report for producing drugs in non-sterile environments. One of their flagship products, feeding tubes for hospital patients, was recalled in the “hundreds of thousands” due to the FDA report.17 This created a “shortage of vitamin B1 for tube-feeding result[ing] in three patient deaths,” tainting LyphoMed’s reputation.18 Undeterred by these setbacks, Kapoor continued running the company until Fujisawa Pharmaceutical, a Japanese pharma company, agreed to buy LyphoMed for nearly $1 billion in 1990. Kapoor made just under $100 million from the sale despite a series of lawsuits with Fujisawa. Fujisawa reluctantly agreed to buy LyphoMed for slightly less than $1 billion after discovering the full extent of its problems with the FDA. Kapoor used the $100 million to continue investing in a series of successful pharmaceutical companies. The majority of his wealth came from Akron, a generic drug manufacturer. Kapoor owned 26% of Akron, worth approximately $890 million by 2016.19 Kapoor’s most ambitious product, however, would not come into being until tragedy struck his personal life.
It was after Kapoor’s wife, Editha, died of breast cancer in 2005 that he decided to make an improved opioid delivery system to manage cancer patients’ pain. After hearing praise for a nitroglycerin spray that treated intense chest pain, Kapoor came up with the idea for putting opioids into a spray product.20 The spray, which he named Subsys, delivers the highly potent synthetic opioid fentanyl to patients in a period of only five minutes, which was three to five times faster than similar opioid medications. After Kapoor’s fentanyl spray Subsys came to fruition in 2012, the FDA approved the product for “breakthrough pain in cancer patients.”21 Many were skeptical that Subsys would be profitable, as only a small number of cancer patients needed to use an opioid as potent as fentanyl. This skepticism proved to be well-founded, as Subsys launched with abysmal sales numbers. Kapoor, unimpressed with the launch, hired a new Vice President of Sales for Insys. This man would ultimately save the company and turn it into one of the country’s fastest-growing pharmaceutical companies.
Alec Burlakoff was a rising star with an incredible talent for sales that he displayed at some of the nation’s largest pharma companies, such as Johnson & Johnson, Eli Lilly, and Cephalon. Kapoor tapped him to be the new Vice President of Sales for Insys soon after the launch of Subsys. Burlakoff made clear in recent interviews why he was hired: to sell dramatically more Subsys by any means possible. He claimed the pressure to succeed was made clear by instructions to “not lose [Kapoor’s] money — or get fired.” Burlakoff went on to admit, “the only way that I knew how to do it, uh, to get that guarantee [of success], is to bribe doctors.”22 The bribery of doctors began soon after Burlakoff’s arrival with the creation of speaker events for doctors that Insys sponsored. Speaker events are commonplace in the medical community to promote new drugs to groups of doctors and physicians. Typically, a few dozen to several hundred doctors gather at these events to hear doctors promote new drug regimes. The Insys speaker events, however, hosted only a handful of doctors, if any, in attendance. A former sales representative for Insys claimed that one speaker event she set up in Chicago had no doctors in attendance other than the speaker. She further claimed that Dr. Madison, the guest speaker for the event, “forge[d] signatures of other doctors that were supposed to be at this dinner program … So that Dr. Madison could be paid.” Dr. Madison received approximately $86,000 from Insys and, in turn, prescribed $1.2 million worth of Subsys for his patients.23 Insys organized thousands of speaker events across the country, massively improving sales among the doctors they paid through these events. Soon after turning to bribery, sales representatives quickly discovered that the doctors who were most willing to take Insys’ money, and in turn prescribe the most Subsys, were the most desperate ones.
Insys was more than happy to have Subsys become a favorite of unscrupulous doctors and addicts alike. Burlakoff stated that Insys sales representatives would target doctors offices “with lines of patients sitting in the parking lot, on the floor drinking Mountain Dew,” which was a common scene at pill mill clinics across the country.24 Kapoor wanted to create a staff of sales representatives complicit with taking advantage of these types of doctors and addicts. To achieve this, he intentionally hired young college grads and those with no experience or formal education in the pharmaceutical industry. This way, the staff was naïve to the damage the product was doing to patients, and as Burlakoff put it, “The key to success [was] the less of the conscience you had, the better.”25 Sales representatives would note doctors’ marital statuses and financial needs to choose who would be the most easily exploited with the speaker money.26
Ironically, the most egregious, and later most damning piece of evidence of Insys’ illegal actions was a simple spreadsheet. Kapoor had it made to include the total speaker money given to each doctor next to the total value of Subsys they prescribed.27 This spreadsheet was clear in its message that Insys expected a return on the money they gave to doctors in the form of Subsys prescriptions. Getting doctors to prescribe Subsys, however, was only half the job of sales representatives. Equally important to the number of prescriptions was the strength of medicine, as higher doses of Subsys brought in more money for Insys but proved more lethal for patients.
Medical titration is the practice of adjusting a patient’s dose of medication to provide maximum benefit with the lowest potential for side effects. Insys, however, seemed to always use the word in reference to increasing dosages, with little regard given to the health and safety of patients. Although Subsys sales increased exponentially following the arrival of Burlakoff and the creation of the speaker series, a relatively low number of doctors were writing the majority of prescriptions. Insys decided that the most effective way of increasing profits was by pressuring existing doctors to increase their patients’ doses of Subsys. A typical one month prescription of 120 units of 200 microgram (mcg) Subsys was worth $6,125 to Insys. This increased dramatically to between $40,875 and $60,000 for a typical month’s supply of the 1600 mcg dosage.28 In emails from former Insys CEO Michael Babich, he expressed the importance of titration, reminding reps they would “make more money the more units and higher dose [they] sell.”29 As part of Kapoor’s initiative to encourage sales over all else, sales representatives for Insys received large bonuses not only for each prescription their doctor’s signed but also for higher dose prescriptions. This ranged from $283 for each 200 mcg prescription to $1,830 for each 1600 mcg prescription. Additionally, monthly competitions awarded $1,500 and $2,000 bonuses to the salespeople who prescribed the most doses over 600 mcg.30
In many company-wide emails, Babich described the work of sales representatives as “selling relief that changes patients lives” and went on to remind Insys employees that “we have heard stories where 800 mcg of Subsys works for some patient[s] … and we have heard others [say] it isn’t strong enough.”31 The company culture, which Babich and others at Insys fostered, taught sales representatives that higher doses meant nothing more than relief for the patient.
However, many of these high doses were given to patients without a developed tolerance to lower doses, which had the potential to cause their death. The FDA warned that Subsys prescribed to “patients not on a chronic regimen of opioids” could result in “lifethreatening respiratory depression and death … at any dose.”32 This did not, however, deter Kapoor and the Insys leadership from pushing titration, and later would cost the lives of thousands of Subsys users. However, Insys still needed a way to claim that these patients without a cancer diagnosis needed Subsys for insurance companies to reimburse them for their prescriptions. To achieve this, Insys simply lied to the insurance companies.
The Insurance Reimbursement Center (IRC) was a subsection of Insys tasked with getting insurance companies to cover as many patient’s prescriptions of Subsys as possible. The concept of reimbursement for medication through insurance is standard in the pharmaceutical industry, as it is the main source of income for drug companies. The standard approval rate for similar opioids was 35-40% of all prescriptions. Kapoor, again wanting to maximize profit, expected closer to a 100% approval rate. However, the vast majority of Subsys prescriptions were for patients without cancer whose doctor prescribed the drug off-label for medical conditions not originally approved by the FDA. Insurance companies almost always refuse to reimburse medicine that is prescribed off-label by doctors. As a workaround, Kapoor had a script developed for the IRC workers to intentionally mislead insurance companies into approving every prescription. When IRC workers called insurance companies to be reimbursed for patients prescribed off-label, they stated that the medication was for “breakthrough pain.” This statement was intentionally ambiguous, and insurance companies assumed it meant the patient had cancer. They would then approve the reimbursement of those off-label prescriptions. A former IRC worker claimed that she truly believed she was helping people, but in hindsight, she realized the insurance fraud she committed contributed to a cycle of addiction and death.33 These illegal tactics by Insys succeeded in deceiving insurance companies, and the creation of the IRC resulted in a massive increase in profits. Through a combination of bribing doctors, pushing higher titration, and scamming insurance companies, Insys made a fortune for its executives and shareholders.
Insys Therapeutics’ stock grabbed the attention of Wall Street as its profits soared after 2013. Reputable brokers were enthusiastically recommending their stock, and it did not disappoint. Insys stock rose from a low of $2.50 at the beginning of 2013 to a high of $44 by mid-2015.34 Burlakoff claimed to have made $8 million from his shares of Insys at its peak.
Kapoor’s shares in Insys increased in value to $650 million by 2016, an almost 300% increase from the IPO price.35 Sales representatives took home handsome paychecks, and investors enjoyed a massive return on investment. At the same time, however, the Department of Justice began noticing a worrying trend: Subsys canisters laying in masses next to addicts who overdosed on Fentanyl. This prompted the U.S. Attorney for Massachusetts to begin crafting a case against Insys after linking hundreds and later thousands of deaths to Subsys in 2015 and 2016. This case, unlike the thousands of pharma cases before it, would apply the RICO Act, a law famously used against organized crime. It would allow the top leadership of Insys all the way to Kapoor to be charged for their illegal and deadly schemes, not only financially, but with jail sentences.
Fighting Organized Crime, No Matter the Collar
The late 1960s saw an unprecedented rise in U.S. urban crime. This included a significant rise in violent crime, which helped drive thousands of middle-class families into the suburbs.36 As police departments began cracking down on this uptick in crime, they managed to prosecute individual criminals with relative ease. The difficulties of taking down large criminal organizations like the mob, however, continued to challenge prosecutors. When the mob was engaged in illegal activities such as selling drugs, gambling, and extortion, the group’s highly sophisticated structure shielded senior members not involved at the street level from prosecution. Additionally, organized crime began infiltrating legitimate business fronts such as “laundry services, liquor and beer distribution, nightclubs, food wholesaling, record manufacturing, [and] the garment industry.”37 Because these business fronts engaged in entirely legal activities, law enforcement often came to dead ends when trying to prosecute the mob. By 1970, Congress began focusing on ways to combat the organized crime problem they estimated was draining billions of dollars from the United States economy.38
In an effort to control organized crime, Congress passed the Organized Crime Control Act of 1970. The act gave law enforcement critical tools to prosecute mob organizations more effectively. A key section of the act established the witness protection program to allow witnesses to cooperate without fear of retaliation from the mob.39 The most important tool the act provided to law enforcement, however, was the Racketeer Influenced and Corrupt Organizations Act, also known as RICO. What made RICO so effective in combating organized crime was its ability to prosecute every individual involved in an enterprise convicted of racketeering. This allowed law enforcement to finally target the leaders of mob groups with criminal convictions, even if they did not commit the crimes themselves.40 The list of activities that the act categorized as racketeering was intentionally left broad and included “murder, kidnapping, arson, gambling, robbery, bribery, extortion, drug dealing, and mail and wire fraud.” Later provisions added securities fraud to the list of racketeering offenses, as fear rose of criminal organizations infiltrating the stock market, and later terrorism in the wake of the September 11th attacks.41
RICO cases require evidence of a minimum of two acts of racketeering over a ten year period for prosecution under the act. If successful, RICO allows for jail sentences of up to 20 years per infraction and increases fines for fraud to up to three times the existing penalties.
Additionally, victims of racketeering can receive restitution for civil damages under the RICO act from corrupt organizations. One of the more controversial portions of RICO, however, was the inclusion of criminal forfeiture in the act. It allows law enforcement to seize the assets and property of those facing RICO prosecution to prevent criminals from bribing witnesses or sending money overseas before trial. Civil libertarians protested this as both infringing on the right to property and threatening to the financial stability of innocent businesses. They also protested the act as creating status crimes, which would make it illegal to simply be part of an organization that may have committed crimes.42 Although these concerns sparked much debate in the Senate, by not singling out one specific type of crime organization, the RICO act was able to pass as part of the larger crime bill.
In the years following the passing of the 1970 Crime Bill, RICO saw only limited use. Law enforcement was not educated on RICO’s uses and continued to struggle with the fight against organized crime. Beginning in the 1980s, however, corrupt organizations and officials were successfully charged under RICO for their crimes. This climaxed with the 1985 Mafia Commission Trial, which boldly went after all five New York mob families in one case. The families had regular meetings between the top leadership, which were called Commission Meetings. After becoming aware of this fact, New York Southern District Attorney Rudy Giuliani used their mafia leaders’ business connections to include twelve mob leaders in a single RICO case. Giuliani’s team successfully argued that the families committed numerous acts of racketeering and linked the group together with Commission meetings to prove they were part of a large criminal enterprise. The case proved to be a resounding success when several mob bosses received 100-year jail sentences. Gradually the mob began to lose its grasp on New York City.43
As RICO gained momentum in the 1980s, important Supreme Court decisions consistently upheld the act’s use no matter the legality of the defendant’s primary business activities. The defense in the Supreme Court Case U.S. v. Turkette (1981) argued the term ‘enterprise’ used to describe organizations that are liable to RICO charges only referred to legitimate business operations and not to illegal activities such as selling illicit drugs. In the landmark case, the court upheld RICO’s use against completely illegal enterprises, whether or not they interfere with legitimate business practices.44 Conversely, Sedima v. Imrex, Co. (1985) upheld RICO’s use against white-collar criminals involved in legal businesses the same as it applies against the mob.45 This interpretation mirrored the perspective of the lawyer and architect of the RICO act Robert Blacky on how RICO should be applied. He argued that the “language [of RICO] says ‘any person.’ There’s nothing about any person who happens to be a member of the mob, or any persona whose name ends in a vowel. The statute doesn’t apply to blue-collar people only, or no-collar people only. It applies to everybody.”46
After several successful RICO convictions against large corporations and the favorable Supreme Court decisions, Giuliani set his sights on the stock market. New types of investments called junk bonds introduced a system to provide capital to companies and individuals with poor credit whose banks would refuse to provide loans. These junk bonds had the potential to provide high yields but were risky due to the high rate of failure of companies that issued these bonds.
Furthermore, junk bonds became a tool to quickly gain massive amounts of capital to finance hostile takeovers and leveraged buyouts of major corporations.47 Although there was nothing inherently illegal about the junk bonds, the system built around it was rife with insider trading and fraud. Giuliani first went after securities company Princeton/Newport Partners with a RICO case for mail fraud in their involvement with junk bonds. Although a federal judge later dismissed the case, the power to withhold Princeton/Newport’s assets drove the company into bankruptcy, sending the rest of the junk bond world into a frenzy. The case brought renewed criticism to the practice of criminal forfeiture within RICO, as Princeton/Newport went out of business after losing access to its capital. Fresh off this victory, Giuliani threatened the same type of RICO case against Drexel Burnham Lambert, one of the leaders in the junk bond market.
After witnessing the bankruptcy of Princeton/Newport who tried to fight the case, Drexel Burnham Lambert agreed to pay a $650 million fine for securities violations, with a stipulation that the government would drop any pending RICO cases against them.48 Giuliani agreed to back off from filing RICO charges against Drexel Burnham Lambert but continued to go after the mastermind behind Drexel Dunham’s junk bond division, Michael Milken. Federal authorities indicted Milken on 98 charges of racketeering under RICO forcing him to take a plea deal of ten years in prison, later reduced to two. Although these companies did commit illegal activities such as tax fraud, many were critical of Giuliani for using RICO to leverage criminal forfeiture against corporations before any court verdicts had been reached. In response, a U.S. Attorney’s memorandum, which advises and sets priorities for federal prosecutors, issued for RICO provided new guidelines that “generally prohibits U.S. Attorneys from bringing RICO or mail fraud charges based on alleged tax violations,” similar to the RICO cases against Princeton/Newport and Drexel Burnham Lambert.49

Despite these restrictions, RICO continued to expand into fighting corruption in the corporate world. A federal judge convicted Louisiana Commissioner of Agriculture and Forestry Gil Dozier of numerous counts of bribery under RICO. The twelve convicted counts of racketeering against him ranged from “$200,000 from Louisiana milk processors … for a promise to lower the price of milk” to “threaten[ing] the owner of an aerial crop dusting service, Roy True, with shutdown unless True paid him $2,000.”51 A judge ultimately sentenced Dozier to eighteen years in prison, two years less than the max sentence, in addition to a $25,000 fine.52 Recently a RICO case has been brought against the International Federation of Association Football (FIFA) for corrupt handling of the World Cup soccer event. Officials in the organization such as David Triesman disclosed information of individuals attempting to buy votes to secure World Cup host nation status and television rights bids.53 In 2015, U.S. federal prosecutors indicted fourteen FIFA officials on RICO charges for laundering $150 million worth of illegal bribes from television networks.54 State Farm found itself in a RICO scandal after journalists uncovered contributions to Lloyd Karmeier’s campaign for a seat in the Illinois Supreme Court. Karmeier won and was a year later part of a 5-4 decision to overturn a multi-billion dollar case against State Farm for failing to use Original Equipment Manufacturer (OEM) parts in car repairs. Some saw the contributions as illegal bribes and attempted to launch a class action RICO case. State Farm, however, ultimately settled for $250 million before the RICO case began in the courts without having to admit guilt.55 One of the first instances of a successful RICO charge for failing to protect consumer health came against the tobacco companies in the early 2000s. The case United States v. Philip Morris successfully argued that tobacco companies deliberately hid evidence of the harm of smoking and marketed their product in a way to mislead consumers about its safety.56
In the past four decades, the expanded uses of RICO allowed the DOJ to consider using the act to tackle the opioid epidemic. Before the Insys case, RICO had only limited use against pharmaceutical companies. The most prominent of these cases is Pfizer Inc. v. Kaiser Foundation Health Plan Inc, which surrounded the off-label prescription of epilepsy drug Neurontin. Pfizer actively promoted these off-label prescriptions of Neurontin to medical professionals for conditions in which it had “effectiveness … no better than a placebo.”57 Because Kaiser, an insurance company, paid for millions of dollars worth of these off-label prescriptions, they used RICO to claim Pfizer committed fraud. After hearing the case, the Supreme Court upheld the $142 million in damages awarded to Kaiser. This same off-label prescription scheme occurred within the Insys Insurance Reimbursement Center. Insys, however, took this a step further by aggressively pursuing doctors to prescribe Subsys with bribes and kickbacks from their sham speaker series. Additionally, Insys’ Subsys medication had a much more lethal effect on its off-label users than a harmless placebo. This made Insys an ideal candidate for a RICO indictment that would be the first case to hold pharmaceutical executives accountable for the opioid epidemic with jail time.58
Justice For Victims of Subsys
On April 24, 2015, an article from investigative reporter Roddy Boyd alerted the public to the first overdose death linked to an Insys product. The first known victim was the previously mentioned Carolyn Markland, who died after taking the drug for back pain due to disc disease. The report, however, also uncovered that the doctor who prescribed her the Subsys medication earned $18,874 through Insys speaker events.59 Soon after Boyd’s report, newspapers began reporting on hundreds of new overdose deaths linked to Subsys. Firefighters, veterans, and mothers all took the drug for off-label pain relief and suffered from Fentanyl addiction leading to an overdose. Addicts like Diana Hampton, a Judge from Nevada, died after resorting to melting down and injecting the leftover fentanyl residue from Subsys canisters.60
As the deaths piled up, the DEA began arresting doctors for prescribing millions of dollars worth of Subsys off-label. Michigan authorities convicted doctor Gavin Awerbuch of receiving over $138,000 in speaker fees from Insys. Dr. Awerbuch in turn prescribed upwards of seven million dollars worth of Subsys to his patients.61 As recently as September 16, 2020, U.S. Attorney Maria Lopez charged pain doctor Steven Chun with violating fraud and anti-kickback laws for receiving over $275,000 from Insys Speaker events. The case also includes Daniel Tondre, an Insys sales representative who set up the events for Dr. Chun, as both men face charges which carry multi-year prison sentences.62 It is likely that litigation against doctors and Insys sales representatives for kickbacks will continue into the foreseeable future.
The Department of Justice began learning of Insys’ illegal activities as early as 2014. In response to insider tips from former Insys sales representatives, the U.S. Attorney’s Office for the District of Massachusetts issued a subpoena of Insys on September 8, 2014.63 This subpoena gave federal prosecutors access to a treasure trove of information on the speaker program and insight into the general company culture. Prosecutors discovered the spreadsheet which tracked the money given to doctors through speaker programs with the amount of Subsys they prescribed. They also found thousands of internal memos, and noticed a particular email from Insys CEO Michael Babich stating that he “thought [Insys] owned the high decile folks,” in reference to his belief that they were immune to any potential federal prosecution.64
After the DOJ gathered enough evidence of illegal bribes and insurance fraud to begin crafting a case, federal prosecutors saw the opportunity to charge Insys executives under the Racketeer Influenced and Corrupt Organizations Act (RICO). This led the U.S. Attorney for Massachusetts Andrew E. Lelling to bring Attorney Fred Wyshak onto the Insys case due to his experience using RICO against the Boston Winter Hill mafia group.65 Wyshak was instrumental in setting up the case against Insys by gathering testimonies from ex-sales and insurance center employees. When remarking on the ethics of Insys compared to the mobsters he previously prosecuted, Wyshak stated, “The mob does to some extent have a code of conduct, and usually they were only physically harming other bad guys … [Insys] didn’t care … Subsys ruined lives, many of them lost their jobs, their families fell apart, some were hallucinating, they all became addicted.”66
On December 8, 2016, the US Attorney’s Office for the District of Massachusetts arrested six Insys Executives on RICO and Racketeering Conspiracy charges. The executives included former CEO Michael L. Babich, former Vice President of Sales Alec Burlakoff, former National Director of Sales Richard M. Simon, former Vice President of Managed Markets Michael J. Gurry, and former Sales Directors Sunrise Lee and Joseph A. Rowan.67 Initially, the DOJ left Insys founder John Kapoor out of the case to continue gathering evidence against him specifically to guarantee a guilty conviction. In response to the RICO case, Kapoor desperately tried to protect the reputation of Insys by denying all wrongdoing and putting new faces in top management positions. This attempt to save face did not, however, save him from prosecution.
Prosecutors soon discovered that Kapoor was part of a daily sales call at 8:30 each morning with Insys executives. The ‘8:30’ calls often discussed the speaker program bribes and the prescription habits of specific doctors receiving the bribes from Insys.68 After collecting ample evidence linking Kapoor to Insys’ bribes and insurance fraud schemes, the DOJ arrested Kapoor on October 26, 2017, as part of the pending RICO conspiracy case against Insys employees.69
After arresting Insys’ top leadership, Attorney Wyshak worked to gather testimony from cooperating defendants in the case. Alec Burlakoff in particular saw little hope in mounting a successful defense and pleaded guilty on November 28, 2018, to the RICO conspiracy and anti-kickback charges.70 Hoping to get a reduced sentence, Burlakoff cooperated with investigators by providing detailed information on the speaker program and admitting to knowingly bribing doctors. In his testimony, Burlakoff stated that Kapoor expected a return in investment of two dollars in Subsys prescriptions for every dollar given to doctors through speaker event bribes. If doctors did not meet this standard, Insys removed them from all future speaking engagements and the bribes that came with it.71 Former CEO Michael Babich pleaded guilty to the RICO charges shortly after Burlakoff on December 26, 2018.72 Babich provided testimony that contained particularly incriminating evidence against his former boss, John Kapoor. He claimed that during a conversation between the two in 2015, just as the DOJ’s investigation intensified, Kapoor asked Babich to leave the company as the “fall man” who would take sole blame for Insys’ illegal activities.7
The hearing for the RICO conspiracy case against Insys executives commenced on January 28, 2019, shortly after the admissions of guilt from Burlakoff and Babich.74 After opening arguments from both sides and questions from the judge, the prosecution showed a video to the courtroom as evidence of Insys’ morally reprehensible company culture. The rap music video was created in 2015 for a National Insys sales meeting and contained lyrics such as, “I love titration, yeah that’s not a problem. I got new patients and I got a lot of ‘em.”75 The video ended with a cameo from Alec Burlakoff in a Subsys medication costume, repeating the song’s chorus in a comedic fashion. Burlakoff admitted that despite the illegal practices the song boasted of, which contributed to thousands of patient deaths, all the sales representatives in attendance of the meeting laughed at the video’s content. He further admitted in recent interviews that this video was made, in effect, to laugh at the grave issue of the opioid epidemic and that “[Insys employees] were all desensitized to what was going on” within the company.76

The Insys rap video played during the 2015 Insys national sales meeting and later during the hearing of Insys Executives on trial for RICO violations7
After several months of arguments, the trial came to a close in April of 2019. On May 2, 2019, a federal jury found John Kapoor and the four remaining Insys executives guilty of RICO conspiracy.78 The conviction was monumental, as it marked the “first successful prosecution of top pharmaceutical executives for crimes related to the prescribing of opioids.”79 The ruling received near-universal acclaim from federal agencies and news organizations alike. U.S. Attorney Andrew E. Lelling, who worked with the prosecution against the Insys defendants, remarked, “Just as we would street-level drug dealers, we will hold pharmaceutical executives responsible for fueling the opioid epidemic by recklessly and illegally distributing these drugs, especially while conspiring to commit racketeering along the way.”80 Sentencing for the Insys executives came eight months after the convictions in January of 2020. Alec Burlakoff and Michael Babich were sentenced to 26 and 30 months in prison respectively. The other four convicted defendants received prison sentences ranging from a year to 33 months. John Kapoor received the harshest sentence of 66 months in prison and restitution amounting to almost $60 million.81 Due to the Covid-19 pandemic, however, the convicted Insys executives have successfully petitioned for their self-surrender dates to be pushed back. As of January 2021, none of the defendants have successfully appealed the aforementioned convictions.
At the same time as former Insys employees faced criminal investigations, Insys itself became engulfed in legal trouble. The DOJ gathered large amounts of information on Insys’ off-label prescription practices, insurance fraud, and bribery of doctors from the RICO investigation. Armed with the damning information, Insys accepted a plea deal from the DOJ on August 8, 2018. The deal settled multiple active civil and criminal investigations with the company for a total payout of $150 million, later increased to $225 million on June 5, 2019.82 The settlement was the final blow to Insys, marking their financial ruin. Several days after the confirmation of the settlement, Insys filed for Chapter 11 Bankruptcy. Without enough capital to pay the $225 million in fines, the courts permitted Insys to sell off its assets to cover a portion of the cost. One of the more notable purchasers of Insys’ assets was Hikma Pharmaceuticals. Hikma purchased “nasal and sublingual spray manufacturing equipment” for $12.2 million, which they plan to use to produce naloxone nasal sprays.83 Naloxone spray, more commonly known by the brand name Narcan, counteracts the effects of drug overdoses, making it a vital weapon in the fight against the opioid epidemic. In a rare glimmer of hope from the dark cloud of the Insys story, it seems that the same machines that once produced a product that killed thousands now might produce a product to save thousands more.
The Next Steps
The successful use of RICO against Insys employees is a signal to the opioid industry that federal prosecutors are willing to criminally prosecute executives who contributed to the over 400,000 lives lost from opioid overdoses. Although the case against Insys was a historic success in holding opioid executives responsible for their actions, the opioid epidemic still rages and requires more attention to curtail the rising death toll.
The next logical target in the DOJ’s efforts to prosecute those responsible for the opioid epidemic is Purdue Pharma executives. Purdue has profited a cumulative $35 billion solely from their drug Oxycontin, which turned millions of Americans into opioid addicts.84 In November, 2020, after years of investigation, Purdue pleaded guilty to one count of fraud and two counts of violating a federal anti-kickback statute. Much like Insys, Purdue participated in programs to funnel money to doctors to encourage the prescription of Oxycontin. Another significant similarity between the two was their complicit attitude towards suspicious doctors who prescribed massive amounts of Oxycontin. Purdue went as far as “[lying] to the Drug Enforcement Administration about steps it had taken to prevent such diversion, fraudulently increasing the amount of its products it was permitted to sell.”85 Unlike Insys, however, no Purdue executives received criminal convictions for their illegal actions. Instead, Purdue Pharma agreed to pay a $3.5 billion criminal fine, $2 billion in criminal forfeiture, and $2.8 billion to resolve civil liability claims, totaling over $8.3 billion.86 Members of the Sackler family, who founded Purdue Pharma and helped push Oxycontin in its early years, paid $255 million to settle civil claims against them.
This case is by no means insignificant; it is, in fact, the highest amount of civil or criminal forfeiture imposed in the history of the pharmaceutical industry. Without any criminal convictions supporting the case, however, many questioned whether this plea deal was appropriate to the destruction that Oxycontin left in its path. Many were particularly appalled at the $225 million settlement with the Sacklers, noting the family’s combined net worth of over $13 billion mostly earned from Oxycontin sales. Some have referenced the fact that the Sacklers withdrew over $10 billion from Purdue over a ten year period as the opioid epidemic was intensifying as proof that they understood their company’s illegal actions.87 Nevertheless, as the coronavirus vaccine dominates the pharmaceutical headlines, it seems the Sacklers are safe from prosecution for the time being.
In closing, the monumental success of the Insys case should be cause for hope in the fight against the opioid epidemic. It seems that the tide may turn in the prosecution of legal opioid manufacturers that will likely result in the continued lowering of overdose deaths linked to prescribed opioids. The fight against illegal opioids and street Fentanyl, however, is making little progress. In 2019, the U.S. had a record number of almost 72,000 overdose deaths, with over half attributable to fentanyl or its derivatives.88 It is clear that stopping the illicit fentanyl trade will require much needed time and effort and should remain the focus of the Drug Enforcement Administration’s efforts. Nevertheless, of the 400,000 Americans who died from opioid overdoses, most became addicted in a similar fashion. Their addiction began after taking prescription opioids such as Oxycontin to deal with injuries or chronic pain. They then sought more potent drugs to feed their addiction, including fentanyl bought on the street or prescribed in the form of Subsys from unscrupulous doctors, leading to their premature deaths. For many, Oxycontin was an entryway into the dark world of addiction that killed thousands and destroyed the lives of millions more who lost their jobs, their families, and their happiness. It is now up to Americans who lost a loved one to an opioid overdose to read the Purdue settlement and decide: is the $8.3 billion an appropriate punishment for the loss of their brother, daughter, mother, or
Bibliography
Asay, Chuck. “RICO Act.” Cartoon. Colorado Springs Gazette Telegraph (Colorado Springs, CO), January 26, 1994. https://cdm15981.contentdm.oclc.org/digital/collection/p15981coll75/id/3446.
Attorney General of Maryland v. Insys Therapeutics, No. 18-028-300480. Accessed November 10, 2020. https://www.marylandattorneygeneral.gov/News%20Documents/insysSOC.pdf.
The Business of Drugs. Season 1, episode 6, “Opioids.” Directed by Jesse Sweet. Aired on Netflix. Accessed November 1, 2020. https://www.netflix.com/watch/80199969?trackId=13752289&tctx=0%2C0%2C9ae95eb bacbb66f03a937c13ec8b1bb90b1ffe9e%3A7fc11732a55d4fb1c646da7a8bf84cf6b0bc45c 9%2C9ae95ebbacbb66f03a937c13ec8b1bb90b1ffe9e%3A7fc11732a55d4fb1c646da7a8b f84cf6b0bc45c9%2Cunknown%2C.
Byers, Barbara A., ed. “John N. Kapoor, PhD ’72.” University at Buffalo. Last modified March 28, 2011. Accessed December 6, 2020. http://www.buffalo.edu/news/releases/2011/03/12424.html.
CDC. “Synthetic Opioid Overdose Data.” Center for Disease Control and Prevention. Last modified March 19, 2020. Accessed January 13, 2021. https://www.cdc.gov/drugoverdose/data/fentanyl.html.
“Understanding the Epidemic.” Center for Disease Control and Prevention. Last modified March 19, 2020. Accessed November 20, 2020. https://www.cdc.gov/drugoverdose/epidemic/index.html.
Cheit, Ross E. “Racketeer Influenced and Corrupt Organizations Act (RICO).” In Encyclopedia of the Supreme Court. N.p.: Facts On File, 2005. online.infobase.com/Auth/Index?aid=15671&itemid=WE52&articleId=167246.
Committee on Homeland Security and Governmental Affairs, Fueling An Epidemic, Report Four, S. Doc., at 159 (2018). Accessed November 3, 2020. https://permanent.fdlp.gov/gpo110284/BoostingFentanylSales.pdf.
Crovitz, Gordon L. “How the RICO Monster Mauled Wall Street.” Notre Dame Law Review Volume 65. Last modified May 2014. Accessed November 17, 2020. https://scholarship.law.nd.edu/cgi/viewcontent.cgi?referer=https://www.google.com/&htt psredir=1&article=2175&context=ndlr.
Davidson, T. (2020). Opioid Epidemic. In J. L. Longe (Ed.), The Gale Encyclopedia of Medicine (6th ed., Vol. 6, pp. 3751-3754). Gale. https://link.gale.com/apps/doc/CX7986601350/HWRC?u=mlin_c_stmarks&sid=HWRC &xid=2fa4c2af
Davidson, Tish, AM, and Meng Li, PhD. “Opioid Epidemic.” In The Gale Encyclopedia of Public Health, 2nd ed., edited by Brigham Narins, 806-810. Vol. 2. Farmington Hills, MI: Gale, 2020. Gale Health and Wellness (accessed October 11, 2020). https://link.gale.com/apps/doc/CX7947900210/HWRC?u=mlin_c_stmarks&sid=HWRC &xid=36d01b73.
Department of Justice Office of Public Affairs. “Opioid Manufacturer Insys Therapeutics Agrees to Enter $225 Million Global Resolution of Criminal and Civil Investigations.” United States Department of Justice. Last modified June 5, 2019. Accessed December 14, 2020. https://www.justice.gov/opa/pr/opioid-manufacturer-insys-therapeutics-agrees-enter-225- million-global-resolution-criminal.
“Opioid Manufacturer Purdue Pharma Pleads Guilty to Fraud and Kickback Conspiracies.” The United States Department of Justice. Last modified November 24, 2020. Accessed December 10, 2020. https://www.justice.gov/opa/pr/opioid-manufacturer-purdue-pharma-pleads-guilty-fraud- and-kickback-conspiracies.
Department of Justice U.S. Attorney’s Office of the District of Connecticut. “APRN Who Received Kickbacks from Insys Therapeutics for Prescribing Fentanyl Spray is Sentenced.” The United States Department of Justice. Accessed January 20, 2021. https://www.justice.gov/usao-ct/pr/aprn-who-received-kickbacks-insys-therapeutics-presc ribing-fentanyl-spray-sentenced.
The Editors of Encyclopaedia Britannica. “Michael Milken.” Encyclopædia Britannica. Last modified June 2020. Accessed November 18, 2020. https://www.britannica.com/biography/Michael-R-Milken#ref260895.
Fear City New York Vs. The Mafia. Season 1, episode 3, “Judgement Day.” Produced by Bernadette Higgins. Aired on Netflix. Accessed December 2, 2020. https://www.netflix.com/watch/81053938?trackId=200257859.
Federal Drug Administration. “HIGHLIGHTS OF PRESCRIBING INFORMATION – Subsys.” fda.gov. Last modified January 2012. Accessed November 10, 2020. https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/202788s000lbl.pdf.
“The fentanyl drug epidemic in North America.” Video, 42:26. YouTube. Posted by DW Documentary, November 6, 2020. Accessed November 11, 2020. https://www.youtube.com/watch?v=TtGpPhd-c7Q&has_verified=1.
“FIFA and the World Cup: Last Week Tonight with John Oliver (HBO).” Video, 13:13. YouTube. Posted by Last Week Tonight, June 9, 2014. Accessed November 16, 2020. https://www.youtube.com/watch?v=DlJEt2KU33I.
“FIFA Corruption Scandal Facts – WMNews Ep. 30.” Video, 13:15. YouTube. Posted by WatchMojo.com, June 10, 2015. Accessed November 16, 2020. https://www.youtube.com/watch?v=cnJIXDu7Vp8.
Frankel, Alison. “Behind $250 million State Farm settlement, a wild tale of dark money in judicial elections.” Reuters. Last modified September 5, 2018. Accessed November 15, 2020. https://www.reuters.com/article/legal-us-otc-darkmoney/behind-250-million-state-farm-settlement-a-wild-tale-of-dark-money-in-judicial-elections-idUSKCN1LL2ZQ.
Freudenheim, Milt. “LYPHOMED SOUGHT BY FUJISAWA.” The New York Times. Last modified August 22, 1989. Accessed December 6, 2020. https://www.nytimes.com/1989/08/22/business/lyphomed-sought-by-fujisawa.html.
Genovese, Michael A. “U.S. Drug Policy.” In Encyclopedia of the American Presidency, Third Edition. N.p.: Facts On File, 2017. online.infobase.com/Auth/Index?aid=15671&itemid=WE52&articleId=208375.
“Health exec with Lake Forest ties is first CEO convicted of opioid racketeering.” Crain’s Chicago Business. Last modified May 2, 2019. Accessed December 6, 2020. https://www.chicagobusiness.com/health-care/health-exec-lake-forest-ties-first-ceo-convi cted-opioid-racketeering.
Herper, Matthew. “An Opioid Spray Showered Billionaire John Kapoor In Riches. Now He’s Feeling The Pain.” Forbes. Last modified October 25, 2016. Accessed November 6, 2020. https://www.forbes.com/sites/matthewherper/2016/10/04/death-kickbacks-and-a-billionaire-the-story-of-a-dangerous-opioid/?sh=2baa35f6e3f9.
H.J. Inc. v. NW Bell Tel. Co., No. 87-1252 (8th Cir. ). Accessed November 16, 2020. https://supreme.justia.com/cases/federal/us/492/229/.
House, Douglas W. “Insys subpoenaed over Subsys commercial practices.” Seeking Alpha. Last modified September 12, 2014. Accessed December 12, 2020. https://seekingalpha.com/news/1981245-insys-subpoenaed-over-subsys-commercial-practices.
https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/pr ess-releases/founder-and-former-chairman-board-insys-therapeutics-sentenced-66-month s-prison.
House of Representatives. “Crime Control Act of 1970.” United States House of Representatives History, Art & Archives. Accessed October 23, 2020. https://history.house.gov/Historical-Highlights/1951-2000/Crime-Control-Act-of-1970/.
“Insys Therapeutics Inc.” The Montley Fool. Accessed November 11, 2020. https://www.fool.com/quote/nasdaq/insys-therapeutics/insy/.
Jacobson, Don. “Purdue Pharma pleads guilty, agrees to pay billions in opioid settlement.” UPI. Last modified October 21, 2020. Accessed January 13, 2021. https://www.upi.com/Top_News/US/2020/10/21/Purdue-Pharma-pleads-guilty-agrees-to- pay-billions-in-opioid-settlement/3831603291661/.
https://www.docketbird.com/court-documents/USA-v-Babich-et-al/JUDGMENT-of-USC A-as-to-Michael-J-Gurry-Richard-M-Simon-Sunrise-Lee-John-Kapoor-re-1274-Notice-o f-Appeal-Conditions-of-Release-1273-Notice-of-Appeal-Conditions-of-Release-1282-No tice-of-Appeal-1281-Notice-of-Appeal-Conditions-of-Release-Defendant-Appe/mad-1:20 16-cr-10343-185341-01381.
Katz, Josh, Abby Goodnough, and Margot Sanger-Katz. “In Shadow of Pandemic, U.S. Drug Overdose Deaths Resurge to Record.” The New York Times. Last modified July 15, 2020. Accessed January 13, 2021. https://www.nytimes.com/interactive/2020/07/15/upshot/drug-overdose-deaths.html.
Keown, Alex. “Trial for Former Insys Therapeutics Founder John Kapoor and Other Executives Set to Begin Today.” BioSpace. Last modified January 28, 2019. Accessed December 14, 2020.
Kounang, Nadia. “Naloxone reverses 93% of overdoses, but many recipients don’t survive a year.” CNN. Last modified October 30, 2017. Accessed November 23, 2020. https://www.cnn.com/2017/10/30/health/naloxone-reversal-success-study/index.html.
Leonard, Thomas M. “Drugs in Modern Latin America.” In Encyclopedia of Modern Latin America (1900 to the Present). N.p.: Facts On File, 2017. online.infobase.com/Auth/Index?aid=15671&itemid=WE53&articleId=244694.
New River Media. “Elliot Currie Interview.” PBS. Accessed November 30, 2020. https://www.pbs.org/fmc/interviews/currie.htm.
“Nine FIFA Officials and Five Corporate Executives Indicted for Racketeering Conspiracy and Corruption.” The United States Department of Justice. Last modified May 27, 2015. Accessed November 16, 2020. https://www.justice.gov/opa/pr/nine-fifa-officials-and-five-corporate-executives-indicted- racketeering-conspiracy-and.
“Opioids, Inc. (full film) | FRONTLINE.” Video, 53:16. YouTube. Posted by FRONTLINE PBS, June 18, 2020. Accessed October 20, 2020. https://www.youtube.com/watch?v=WvCld2vKug8&feature=emb_logo.
“Opium, 1800–1950.” In Encyclopedia of Modern China, edited by David Pong, 57-59. Vol. 3. Detroit, MI: Charles Scribner’s Sons, 2009. Gale In Context: World History (accessed October 12, 2020). https://link.gale.com/apps/doc/CX1837900434/WHIC?u=mlin_c_stmarks&sid=WHIC& xid=044d085f.
Pacenti, John. “Veteran, would-be lawyer, mom left dead or addicted after Subsys.” The Palm Beach Post. Last modified April 4, 2018. Accessed December 10, 2020. https://www.palmbeachpost.com/news/20180404/veteran-would-be-lawyer-mom-left-dea d-or-addicted-after-subsys.
Palmer, Eric. “Hikma picks up Insys naloxone spray, equipment in $12M bankruptcy buy.” Fierce Pharma. Last modified August 20, 2019. Accessed October 29, 2020. https://www.fiercepharma.com/manufacturing/hhikma-picks-up-insys-naloxone-spray-an d-equipment-12m-bankruptcy-buy.
Pavlo, Walter. “Once Meant To Nail Mobsters, RICO Sees Resurgence In Civil Cases In 2018.” Forbes. Last modified October 31, 2018. Accessed November 15, 2020. https://www.forbes.com/sites/walterpavlo/2018/10/31/once-meant-to-nail-mobsters-rico-s ees-resurgence-in-civil-cases-in-2018/?sh=4cb86c222421.
“Pharma execs used strip clubs, broke FDA laws to boost opioid sales.” Video, 27:04. YouTube. Posted by 60 Minutes, June 23, 2020. Accessed October 26, 2020. https://www.youtube.com/watch?v=G1jLVP156_E.
Raymond, Nate. “Former Insys CEO Babich Pleads Guilty to Opioid Prescription Bribe Scheme.” Insurance Journal. Last modified December 27, 2018. Accessed December 14, 2020. https://www.insurancejournal.com/news/national/2018/12/27/512860.htm27.
“Insys to pay $150 million to settle U.S. opioid kickback probe.” Reuters. Last modified August 8, 2018. Accessed December 14, 2020. https://www.reuters.com/article/us-insys-opioids/insys-to-pay-150-million-to-settle-u-s-o pioid-kickback-probe-idUSKBN1KT1G5.
Reuters Staff. “Sacklers withdrew over $10 billion in a decade from Purdue Pharma: New York Times.” Reuters. Last modified December 16, 2019. Accessed January 13, 2021. https://www.reuters.com/article/us-purduepharma-bankruptcy-audit/sacklers-withdrew-ov er-10-billion-in-a-decade-from-purdue-pharma-new-york-times-idUSKBN1YL060.
“60 Minutes Archives: Buying fentanyl online from China.” Video, 12:27. YouTube. Posted by 60 Minutes, December 13, 2020. Accessed December 14, 2020. https://www.youtube.com/watch?v=mkyLQEnFggE.
Spector, Mike, and Tom Hals. “Exclusive: OxyContin maker Purdue is ‘Pharma Co X’ in U.S. opioid kickback probe – sources.” Reuters. Last modified January 28, 2020. Accessed November 1, 2020. https://www.reuters.com/article/us-purdue-pharma-investigation-opioids-e/exclusive-oxycontin-maker-purdue-is-pharma-co-x-in-us-opioid-kickback-probe-sources-idUSKBN1Z R2RY.
“Subsys Rap Video Created by Insys Pharmaceuticals (More info in description).” Video, 3:53. YouTube. https://www.youtube.com/watch?v=mtwFZwjCSTE&t=79s.
Sullivan, Thomas. “Supreme Court Rejection of Pfizer’s Request for RICO Off Label Review: Could Open Floodgate of Cases.” Policy & Medicine. Last modified May 6, 2018. Accessed December 3, 2020. https://www.policymed.com/2014/01/supreme-court-rejection-of-pfizers-request-for-rico- off-label-review-could-open-floodgate-of-cases.html.
Tong, William. “UNSEALED OPIOID COMPLAINT– ATTORNEY GENERAL TONG RELEASES NEW SHOCKING EMAILS FROM PURDUE’S RICHARD SACKLER.”The Office of Attorney General William Tong. Last modified May 6, 2019. Accessed November 1, 2020. https://portal.ct.gov/AG/Press-Releases/2019-Press-Releases/ATTORNEY-GENERAL-T ONG-RELEASES-NEW-SHOCKING-EMAILS-FROM-RICHARD-SACKLER.
“Too big to prosecute.” Video, 15:11. YouTube. Posted by 60 Minutes, September 30, 2018. Accessed November 6, 2020. https://www.youtube.com/watch?v=R55POy5YJ5Q.
The United States Department of Justice, comp. The False Claims Act: A Primer. Accessed October 29, 2020. https://www.justice.gov/sites/default/files/civil/legacy/2011/04/22/C-FRAUDS_FCA_Pri mer.pdf.
“109. RICO Charges.” DOJ. Last modified January 22, 2020. Accessed December 1, 2020. https://www.justice.gov/archives/jm/criminal-resource-manual-109-rico-charges.
United States Department of Justice. https://www.justice.gov/usao-ma/pr/former-ceo-insys-therapeutics-sentenced-racketeerin g-scheme.
United States Drug Enforcement Administration. “Drug Scheduling.” DEA. Accessed November 8, 2020. https://www.dea.gov/drug-scheduling.
“United States of America, Plaintiff-appellee, v. Gilbert L. Dozier, Defendant-appellant, 707 F.2d 862 (5th Cir. 1983).” Justia US Law. Accessed December 3, 2020. https://law.justia.com/cases/federal/appellate-courts/F2/707/862/230224/. United States v. L Dozier, No. 80-3927 (5th Cir. ). Accessed December 3, 2020. https://openjurist.org/672/f2d/531/united-states-v-l-dozier.
United States v. Michael Babich, Alec Burlakoff, Richard Simon, Sunrise Lee, Joseph Rowan, and Michael Gurry, John Kapoor, H.D. (Mass. ). Accessed October 16, 2020. https://www.justice.gov/usao-ma/victim-and-witness-assistance-program/united-states-v- michael-babich-alec-burlakoff-richard-simon-sunrise-lee-joseph-rowan-and.
United States v. Philip Morris, No. 99-2496. Accessed December 3, 2020. https://www.publichealthlawcenter.org/topics/commercial-tobacco-control/commercial-tobacco-control-litigation/united-states-v-philip.
U.S. Attorney’s Office District of Massachusetts. “Founder and Four Executives of Insys Therapeutics Convicted of Racketeering Conspiracy.” United States Department of Justice. Last modified May 2, 2019. Accessed December 14, 2020. http://www.justice.gov/usao-ma/pr/founder-and-four-executives-insys-therapeutics-convi cted-racketeering-conspiracy.
U.S. Attorney’s Office Middle District of Florida. “Sarasota Pain Doctor And Former Insys Sales Representative Charged In Health Care Fraud Kickback Conspiracy.” The United States Department of Justice. Last modified September 16, 2020. Accessed December 11, 2020. https://www.justice.gov/usao-mdfl/pr/sarasota-pain-doctor-and-former-insys-sales-repres entative-charged-health-care-fraud.
U.S. Attorney’s Office of the District of Massachusetts. “Former Vice President of Insys Pharmaceuticals Pleads Guilty to Racketeering Scheme.” The United States Department of Justice. Last modified November 28, 2018. Accessed December 14, 2020. https://www.justice.gov/usao-ma/pr/former-vice-president-insys-pharmaceuticals-pleads- guilty-racketeering-scheme.
“Founder and Former Chairman of the Board of Insys Therapeutics Sentenced to 66 Months in Prison.” United States Department of Justice. Last modified January 23, 2020. Accessed December 17, 2020. https://www.justice.gov/usao-ma/pr/founder-and-former-chairman-board-insys-therapeuti cs-sentenced-66-months-prison.
“Founder and Owner of Pharmaceutical Company Insys Arrested and Charged with Racketeering.” The United States Department of Justice. Last modified October 26, 2017. Accessed December 14, 2020. https://www.justice.gov/usao-ma/pr/founder-and-owner-pharmaceutical-company-insys-arrested-and-charged-racketeering.
“Pharmaceutical Executives Charged in Racketeering Scheme.” United States Department of Justice. Last modified December 8, 2016. Accessed December 13, 2020. https://www.justice.gov/usao-ma/pr/pharmaceutical-executives-charged-racketeering-sch eme.
“The Whistleblower.” Video, 27:19. YouTube. Posted by 60 Minutes, September 30, 2018. Accessed November 6, 2020. https://www.youtube.com/watch?v=Hd-43K-rdQA.
Wiland, Harry, Andrew Kolodny, Georges Benjamin, and Peter Segall. Do No Harm: The Opioid Epidemic. Nashville: Turner Publishing Company, 2020.
Zorea, Aharon W. “Racketeer Influenced and Corrupt Organizations (RICO) Act.” Contemporary United States, Third Edition. Facts On File, 2017. Accessed October 23, 2020. online.infobase.com/Auth/Index?aid=15671&itemid=WE52&articleId=209332.
Zorea, Aharon W. “Narcotics.” In Contemporary United States, Third Edition. N.p.: Facts On File, 2017. online.infobase.com/Auth/Index?aid=15671&itemid=WE52&articleId=209956.
“Racketeer Influenced and Corrupt Organizations (RICO) Act.” In Contemporary United States, Third Edition. N.p.: Facts On File, 2017. online.infobase.com/Auth/Index?aid=15671&itemid=WE52&articleId=209332.


