By Nathan Cunningham, VI Form
Development of the Bohr Model of the Atom
 Around the turn of the 20th century, physicists became increasingly focused on examining the most miniscule of physical interactions. The concept of quanta—the smallest piece of something, e.g. the American cent—led into the evolution of quantum mechanics, the fundamental study of the components of matter and light. This piece will explore the various experiments, discoveries, and theories at the quantum level of world interactions which led up to the development of the Bohr model of the atom.
Around the turn of the 20th century, physicists became increasingly focused on examining the most miniscule of physical interactions. The concept of quanta—the smallest piece of something, e.g. the American cent—led into the evolution of quantum mechanics, the fundamental study of the components of matter and light. This piece will explore the various experiments, discoveries, and theories at the quantum level of world interactions which led up to the development of the Bohr model of the atom.
Wilhelm Conrad Röntgen was an influential figure in quantum mechanics. As one of the initial scientists to practice with electric discharge in gases, he was a specialist in light phenomena, and he is responsible for one of the greatest discoveries in history: the discovery of the X-ray. He frequently experimented with Crookes tubes, partially-evacuated glass tubes with a positive anode and negative cathode at opposite ends. When a current was run through the electrodes, a glow was produced in the tube. On November 8th, 1895 when Röntgen ran a high-voltage current through one of his tubes, he noticed the same glow produced in the tube on a screen covered with barium platinum cyanide which he held at a distance (“Wilhelm Röntgen”). Röntgen then covered the tube in opaque black paper and saw that the glow persisted through the paper and onto the screen. What Röntgen was observing was radiation created when the high-voltage current ran from the cathode and struck the anode. The current was such a high voltage that the anode was struck with enough energy to emit the high-energy, short wavelength X-rays. The glow of radiation could only be produced by a very high energy source. A property of X-rays that was never observed before was the ability of the radiation to penetrate different materials to different degrees. X-rays are a great use in medical practices because they can go through human tissue easily, allowing us to produce images of broken bones. X-rays fall on the right side of the light spectrum which will be further explained through the development of quantum physics. The discovery of X-rays was not directly influential on Niels Bohr’s development of his model of the atom, but X-rays opened the path to other discoveries and theories in the behavior of light.
Before 1897, it was believed that the atom was the elementary quanta of all matter (Einstein, 1938). J.J. Thomson changed that belief with his discovery of the electron. Thomson, much like Röntgen with Crookes tubes, experimented with cathode ray tubes (glass tubes filled with a low pressure gas). When a current was run from the negative cathode to the positive anode through the tube, a glow became present in the tube. Thomson, aiming to learn more about the actual cause of the glow, manipulated the rays from the cathode with both electric and magnetic fields, and he measured the deflection of the rays in relation to the strength of the fields and discovered that the rays had a charge to mass ratio of 1.8*1011 coulombs per kilogram (nobelprize.org). After performing several other experiments and deflecting the rays by different amounts each time, Thomson noticed that the charge to mass ratio stayed the same. He concluded that the rays must be made from particles with this distinct charge to mass ratio, and that these particles were components of all matter. In 1904, Thomson formed his plum-pudding model of the atom: the atom consists of negative electrons floating in a positive fluid. This model led to Ernest Rutherford’s famous gold-foil experiment, where he shot positively-charged alpha particles at an extremely thin piece of gold foil and observed that some had deflected and bounced back. This revealed that the atom had a densely packed center mass of positive charge which Rutherford called the nucleus. In 1911, Rutherford then created his planetary model of the atom, where electrons all orbit the nucleus at a great distance like planets around the sun (Kaplan, 2015, p. 262). These two atomic models were influential in Bohr’s thinking, and the discovery of the electron was momentous in the analysis of quantum phenomena.
The quantization of light—the division of light into the smallest unit possible—largely began as an inkling of Isaac Newton’s. Newton believed that light was a shower of tiny pieces he called corpuscles, but when this model failed to explain the diffraction, interference, and bending of light, Christiaan Huygens’s wave model of light became truth. Then a new phenomenon of light was observed: “Homogeneous light, such as violet light, which is, as we know, light of a definite wave-length, is impinging on a metal surface. The light extracts electrons from the metal… This extraction of electrons by light falling upon metal is called the photoelectric effect,” (Einstein, 1938, p. 258). The photoelectric effect was first observed by Heinrich Hertz when he shined ultraviolet light on a piece of zinc metal and noticed that the metal became positively charged, and, although Hertz didn’t know it at the time, we now know that the light was removing electrons from the zinc (Kaplan, 251). The photoelectric effect could only be explained as if light was made up of tiny, individual bits, each of which contained a specific amount of energy. Max Planck was the first to theorize about the quanta of light, the photon, resolving that the energy of a photon was proportional to its frequency. Albert Einstein refined Planck’s idea to further explain the photoelectric effect: one photon will remove one electron but the photon needs enough energy to first extract the electron and then transfer some kinetic energy so the electron can escape (Kaplan, 2015, p. 252). Different metals have different threshold frequencies which correspond to the minimum energy of the light needed to produce the photoelectric effect. Light with greater energy will have a shorter wavelength and a higher frequency. This links the color of the light with the energy of the photons it emits, as the wavelength of light increases as you go from left to right on the spectrum, which will be discussed in the next section The quantization of light and the connection between photons, energy, and color are all crucial ideas which help Bohr postulate about the atom and form his model.
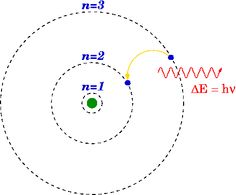
The light spectrum is where Bohr was able to make his greatest step towards his model of the atom. Seen on the spectrum below, waves that are categorized as “light” are depicted from left to right from lowest energy to highest energy. 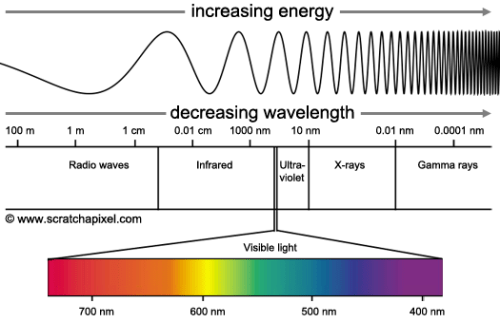
The visible light spectrum makes up only a very small portion of the entire light spectrum, and it is shown that X-rays have much shorter wavelength and higher energy than any visible light. Much like Röntgen’s experiments with Crookes tubes, a bright and distinct glow emanates when a current is run through a gas-filled tube. Through experimentation with the excitement of gases, atomic spectra were discovered by many scientists who observed this phenomenon; different elements, when excited, produced different colored light. One scientist in particular, Niels Bohr, was keenly intrigued by observing excited gas through a spectrometer. He inspected excited hydrogen gas and noticed that the spectrometer diffracted the light into several colors in the visible light spectrum, however, the spectrum was discontinuous with colors showing in bright, separated lines (Kaplan, 2015, p. 262). This discontinuous spectrum was commonly observed, but Bohr, with a stroke of brilliance, connected the discontinuous spectrum of excited hydrogen gas and the structure of the atom. Much like Rutherford’s planetary model of the atom, Bohr proposed that the electrons of an atom orbit the nucleus, however, he suggested that they orbit at specific distances, and he used the discontinuous spectrum to support this. Linking the energy of light with the energy of the electrons in orbit, Bohr proposed that when the atoms of the hydrogen gas became excited by the current, the electrons noticed an increase in energy and changed orbits; the spectrum was discontinuous because the orbits of the electrons were discontinuous, and the electrons emitted light when changing orbits and changing energy.
Each of the experiments and theories outlined were influential to Bohr’s learning and thinking, and all played a significant role in the development of the Bohr model of the atom. Although the practice of experimentation with light phenomena greatly predates Hertz, Röntgen, Thomson, Planck, and Bohr, these five collectively amounted for some of the greatest lab discoveries in the history of the world. Quantum mechanics would not be as far along today if where not for the progress made by scientists around the turn of the 20th century. Bohr, as brilliant as he was, was simply a benefactor of the outstanding work done before him, and his model is the result of thousands of great minds coming together.
 Nathan Cunningham is a VI Former from Bow, New Hampshire, who lives in Coe House. He enjoys math and science and plays varsity football and hockey.
Nathan Cunningham is a VI Former from Bow, New Hampshire, who lives in Coe House. He enjoys math and science and plays varsity football and hockey.
Bibliography
Abramowitz, M., Neaves, S. H., & Davidson, M. W. (2015, November 13). The
Frequency and Wavelength of Light. Retrieved March 10, 2016, from Molecular
Expressions website: https://micro.magnet.fsu.edu/optics/lightandcolor/
frequency.html
Einstein, A., & Infeld, L. (1938). The Evolution of Physics. New York, NY:
Touchstone.
The Electron Discovery Experiment. (2016). Retrieved March 10, 2016, from
Nobelprize.org website: http://www.nobelprize.org/educational/physics/
vacuum/experiment-1.html
Fitzpatrick, R. (2007, July 14). Huygens’ principle. Retrieved March 10, 2016,
from http://farside.ph.utexas.edu/teaching/302l/lectures/node150.html
In-Text Citations: The Basics. (2016). Retrieved March 10, 2016, from Purdue
Online Writing Lab website: https://owl.english.purdue.edu/owl/resource/560/02/
Joseph John Thomson. (2016). Retrieved March 10, 2016, from Chemical Heritage
Foundation website: http://www.chemheritage.org/discover/online-resources/
chemistry-in-history/themes/atomic-and-nuclear-structure/thomson.aspx
A Look Inside the Atom. (2016). Retrieved March 10, 2016, from American
Institute of Physics website: https://www.aip.org/history/exhibits/electron/
jjhome.htm
SAT Subject Test Physics. (2015). New York, NY: Kaplan Publishing.
This Month in Physics History November 8, 1895: Roentgen’s Discovery of X-Rays.
(2015, November). Retrieved March 10, 2016, from American Physical Society
website: https://www.aps.org/publications/apsnews/200111/history.cfm
Tuckerman, M. E. (2011, September 13). J.J. Thomson’s experiment and the
charge-to-mass ratio of the electron. Retrieved March 10, 2016, from
https://www.nyu.edu/classes/tuckerman/adv.chem/lectures/lecture_3/node1.html
Wilhelm Röntgen. (2016). Retrieved March 10, 2016, from Famous Scientists
website: http://www.famousscientists.org/wilhelm-rontgen/
X-Rays by Wilhelm Conrad Röntgen (Milestones of Science) [Lecture transcript].
(2010, March 2). Retrieved March 10, 2016, from YouTube website:
https://www.youtube.com/watch?v=I6Fu0GcLhVE


